-
Fil d’actualités
- EXPLORER
-
Pages
-
Groupes
-
Evènements
-
Blogs
-
Offres
-
Emplois
-
Courses
How to Document Immediate Use Steam Sterilization (IUSS) Cycles for Compliance
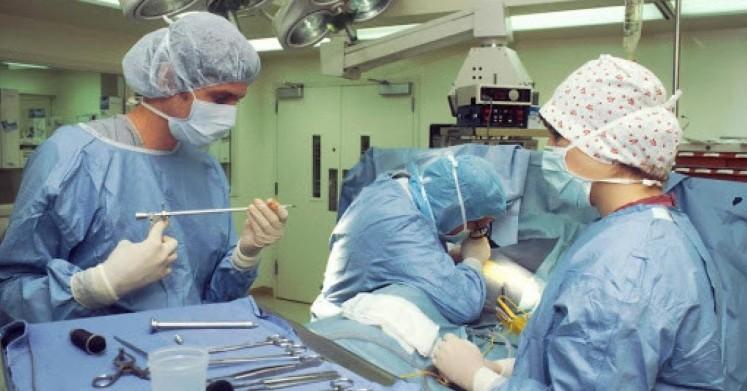
In the high-stakes environment of the operating room, the need for surgical instrumentation can sometimes exceed the available supply, leading to the use of Immediate Use Steam Sterilization (IUSS). Formerly known as "flash sterilization," IUSS is a process designed for the rapid sterilization of instruments that are needed for immediate patient use. However, because this process bypasses certain drying phases and long-term storage protocols, regulatory bodies like The Joint Commission and CMS maintain incredibly strict documentation requirements. For any healthcare facility, failing to document these cycles correctly can lead to severe compliance penalties and, more importantly, compromised patient safety.
Defining the Justification for IUSS Usage
The first and perhaps most critical step in documenting an IUSS cycle for compliance is the "statement of necessity." Regulatory inspectors look for clear evidence that IUSS was used only in urgent situations and not as a substitute for an inadequate instrument inventory. To remain compliant, the documentation must specify exactly why the item could not be processed via the standard terminal sterilization method. Was it a one-of-a-kind instrument that was accidentally contaminated? Was there an unexpected change in the surgical procedure? Simply stating "needed for surgery" is often insufficient during an audit. Technicians must be trained to record the specific clinical rationale that justifies the deviation from standard protocol.
Capturing Essential Cycle Parameters and Identifiers
Once the justification is established, the technical documentation must capture the granular details of the sterilization cycle itself. This includes the unique identifier for the sterilizer used, the specific cycle type (such as gravity-displacement or pre-vacuum), and the temperature and duration of the exposure phase. Compliance requires that the physical monitor printout or digital log be reviewed and initialed by the operator to verify that all parameters were met.
Furthermore, the documentation must identify the specific instruments processed in that batch. If a patient develops a post-operative infection, the hospital must be able to trace exactly which tools were sterilized via IUSS for that specific case. This level of meticulous record-keeping is drilled into students during their sterile processing technician course, ensuring they understand the legal weight that these logs carry in a court of law or during a state inspection.
Tracking the Biological and Chemical Indicators
No IUSS documentation is complete without the inclusion of biological and chemical indicator results. For IUSS, a Class 5 or Class 6 chemical integrator is typically required within the sterilization container to provide immediate visual confirmation that the necessary conditions for sterilization were achieved. The results of these indicators must be recorded in the permanent log. While daily biological testing is standard for terminal sterilization, some facilities require a biological indicator with every IUSS load containing an implantable device, which must then be tracked until the results are confirmed. The complexity of managing these different indicator types and their respective incubation periods is a sophisticated task.
Patient-Linkage and Post-Sterilization Transport
A common point of failure in IUSS compliance is the failure to link the sterilization cycle to a specific patient. To meet AAMI and AORN standards, the documentation must include the patient's name or unique medical record number. This creates a "closed loop" of accountability. Additionally, the documentation should reflect the method of aseptic transport from the sterilizer to the point of use. Since IUSS items are often processed without the heavy wraps used in terminal sterilization, they are highly vulnerable to re-contamination during transport. Recording the time the item left the sterilizer and the initials of the person who delivered it to the sterile field provides a complete chain of custody.
Managing the Quality Assurance and Audit Trail
Documentation for IUSS doesn't end when the surgery is over; it must be incorporated into a larger quality assurance program. Facilities are required to perform regular "IUSS audits" to identify trends. For instance, if one surgical specialty is consistently relying on IUSS for the same set of instruments, it indicates a need for capital investment in more instrument trays rather than a continued reliance on rapid sterilization. Compliance officers look for evidence that the leadership team is reviewing these logs to drive down the frequency of IUSS usage.
Conclusion: The Professional Responsibility of Record-Keeping
Ultimately, the proper documentation of IUSS cycles is a testament to the professionalism of the sterile processing department. It is a rigorous, repetitive, but absolutely essential task that protects the most vulnerable members of the healthcare system: the patients on the operating table. When a technician signs off on an IUSS log, they are making a professional guarantee that the instruments are safe for use. This level of accountability requires a deep sense of ethics and a mastery of technical protocols, both of which are fostered through dedicated education.
- Art
- Causes
- Crafts
- Dance
- Drinks
- Film
- Fitness
- Food
- Jeux
- Gardening
- Health
- Domicile
- Literature
- Music
- Networking
- Autre
- Party
- Religion
- Shopping
- Sports
- Theater
- Wellness


